Flexible Ureteroscopy
 Video
Video
Flex XC1 Testimonial: Dr. Michael Lipkin, Duke University
Dr. Lipkin shares his first impressions on the Flex XC1 Single Use Ureteroscope, how he sees it playing a role in his practice, and additional insights into the ergonomic and operational benefits of a single use solution.
 Video
Video
Flex XC1 Testimonial: Dr. Sri Sivalingam, Cleveland Clinic
Dr. Sivalingam shares his first impressions on the Flex XC1 Single Use Ureteroscope, how he sees it playing a role in his practice, and additional insights into the benefits of having choices in your endoscope offering.
 Video
Video
Flex XC1 Testimonial: Dr. Amy Krambeck, Northwestern University
Dr. Krambeck shares her first impressions on the Flex XC1 Single Use Ureteroscope, how it compares to her reusable scopes, and how she sees it playing a role in her practice.
 Document
Document
Choice adds flexibility in all aspects of your patient care. Clinical, Operational, and Financial Choices to meet your unique needs – All from KARL STORZ.
 Video
Video
NEW: Flex XC1 Single Use Ureteroscope
Choose Convenience without Compromise! The new Flex XC1 single use ureteroscope offers best in class image quality in a convenient single use package.
 Document
Document
NEW: Flex XC1 Single Use Ureteroscope Specifications
HIGHLIGHTS • CMOS Chip • Integrated LED light source • 105° Angle of view • 0° Direction of view • 270° Deflection (up and down) • 700 mm Working length • 3.5 Fr. Inner channel • 3 O’clock position working channel • 9 Fr. Sheath size • 9 Fr. Tapered dis
 Video
Video
(Video that highlights the 3 different scopes) Flexibility to choose the right scope for each patient and procedure. Reusable Digital and Fiber Scopes as well as single use – All from KARL STORZ.
Minimally Invasive PCNL: MIP
 Video
Video
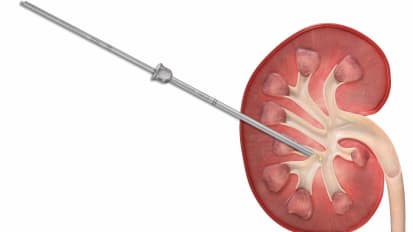
Minimally Invasive PCNL Technique and Demo
Minimally Invasive PCNL (MIP) is more than a mini-perc. This system takes advantage of single-step dilation and the vacuum effect to remove stones with minimal trauma and faster procedure times. Send your patients home with just a bandage.
 Video
Video
Minimally Invasive PCNL Surgery Technique
Minimally invasive percutaneous nephrolithotomy for stone removal offers advantages for surgeon and patient alike. Using a live case study, this 14-min. video explains the whole procedure, from equipment to technique.
 Video
Video
Renal Access for PCNL: Step-by-Step Guide
This 18-min. instructional video provides a step-by-step guide to renal access for percutaneous nephrolithotomy. It was prepared by Julio Davalos, MD, of Chesapeake Urology Associates and the University of Maryland.
 Video
Video
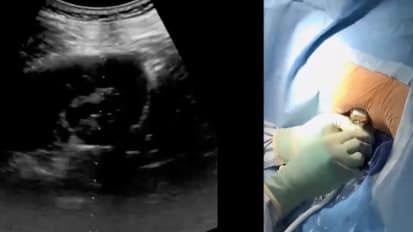
Ultrasound Guidance for Mini PCNL
Fluoro-guided access is still the norm for minimally invasive PCNL. Is ultrasound a better option? In this master class, Dr. Roger Sur from UC San Diego shares his experience using ultrasound, with practical tips for success.
 Video
Video
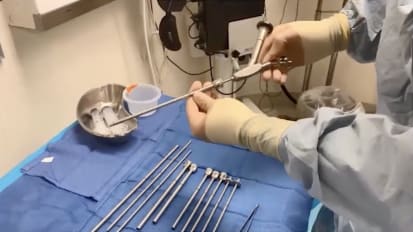
Mini PCNL Instrumentation and Demo
Dr. Seth Bechis of UC San Diego introduces the reusable instrument set needed to perform minimally invasive PCNL on an outpatient basis and demonstrates procedural steps, including the vacuum effect.
 Video
Video
Minimally Invasive PCNL vs. Ureteroscopy
Dr. Sri Sivalingam of the Cleveland Clinic compares minimally invasive PCNL and ureteroscopy, especially for treating stones from 1 to 2 cm. Stone-free rates, bleeding risk, ergonomics, and tubeless options are discussed.
 Video
Video
Mini PCNL in the Outpatient Setting
Is minimally invasive PCNL suitable for outpatient settings? In this master class, Dr. Julio Davalos of Chesapeake Urology gives practical tips and considerations for transitioning MIP cases from the hospital to the ASC.
Reimbursement Resources
 Document
Document
Transitional Pass-Through Payment Overview
Overview to the new TPT payment implemented by CMS on Jan. 1, 2023 for single-use endoscopes including the KARL STORZ Flex XC1 ureteroscope
 Document
Document
2023 guide to stone management procedural coding and reimbursement for inpatient, outpatient and ASC settings.
Flexible Cystoscopy
 Video
Video
Enabling Anywhere Care: Office/Clinic/ASC
TELE PACK+ Compact Endoscopy and the C-VIEW Digital Cystoscope are simple and cost-effective flexible cystoscopy solutions for the Urology office/clinic/ASC
 Video
Video
Simple to Use, versatile, and cost effective at 1/3 cost of a typical OR Video Tower
 Video
Video
C-VIEW Digital Cystoscope - Cystoscopy Simplified
Simple to use, simple to maintain, and cost-effective. Save $100/case over single-use options (includes reprocessing and repair exchange service costs).
Care & Handling
 Video
Video
Flexible Ureteroscope Care and Maintenance
Maximize your service life and protect your investment by following these basic tips concerning setup, cleaning, and transport of your flexible ureteroscope. Proper care can help lower your total cost of ownership.
 Document
Document
E-Class Brochure - Flexible Endoscope
You and your patients deserve endoscopes that meet the same high quality standards and perform as reliably as brand new endoscopes after every service. KARL STORZ E-Class® exchange program was built on that philosophy.
Bladder Cancer
 Document
Document
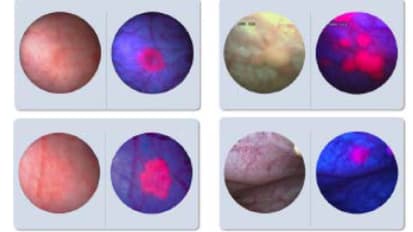
Value Analysis Brief KARL STORZ Blue Light Cystoscopy with Cysview® (BLCC) System*
Value Analysis Brief KARL STORZ Blue Light Cystoscopy with Cysview® (BLCC) System
 Document
Document
Blue Light Cystoscopy Clinical Advantages
Thomas Cangiano, MD, Board-certified urologist at AdventHealth Altamonte Springs, FL, describes his initial experiences and observations using Blue Light Cystoscopy with Cysview® (BLCC) on his patients.
 Video
Video
Blue Light Cystoscopy Patient Case Study
At 28 years old, Mary Beth Ballard received a bladder cancer diagnosis that would change her life. Doctors at Vanderbilt University Medical Center were able to treat her using blue-light cystoscopy. This is her story.
Energy
 Video
Video
Surgical Generator for Bipolar Resection
The UH 400 surgical generator provides high-frequency current for endoscopic cutting, coagulation, and dissection. Pair it with our true bipolar electrodes, designed to deliver maximum control with minimal energy.
 Video
Video
Bipolar Electrodes for Urology Surgery
Our true bipolar electrodes come in various styles to meet diverse surgical needs. Watch how precisely they perform during endoscopic bladder dome and posterior wall resections, treatment of papillary lesions, and more.
Thulium Fiber Laser
 Document
Document
On-on-One Consulting: Thulium Fiber Laser
KARL STORZ Preceptorship program is designed to support your clinical practice questions via one-on-one consulting. Check out our KARL STORZ Preceptor Info Sheet to see how you can get connected.
 Document
Document
Detailed overview of KARL STORZ MultiLASE TFL Specifications; including, but not limited to Laser Classification, Operating Parameters, Dimensions, and Energy Requirements.
 Document
Document
Overview of KARL STORZ newly launched Thulium Fiber Laser, MultiLASE TFL. High-level specs, part numbers and accessories can be found in this document.
 Document
Document
On-Site Endoscopic Laser Specialist
Overview of KARL STORZ On-Site Endoscopic Program to support your laser operation, reprocessing and set-up needs. This program was developed to help our customers get the support they deserve.
 Video
Video
MultiLASE TFL Inservice & Setup Guide
This instructional video is for anyone who uses or sets up the Karl Storz MultiLASE TFL. Learn how to properly set up the laser and adjust key settings to ensure a safe and smooth procedure.
The products described in the videos, images, and documents on this site have been approved authorized by the company for use in the United States only. The materials on this site are provided exclusively as an educational service for medical professionals and do not necessarily reflect the views of KARL STORZ United States. The decision to apply any of the information on this site is at the sole discretion and choice of qualified medical personnel, who are advised to verify the applicability and the suitability for any particular situation. KARL STORZ cannot be held is not responsible for the misuse of any information on this site.